
Instead, the electrons orbit around the nucleus of the atom, which is constantly moving. The electrons would orbit around the nucleus of the atom.Ītomic Model Theory is the idea that the electrons orbiting the atom don’t orbit around a stationary nucleus like they were on the earth in a solar system. Bohr’s atomic model had a nucleus with a certain number of positively charged particles that were held together by negatively charged particles. Niels Bohr was a Danish physicist who had a theory about atoms that he called the “atomic model”. Niels Bohr Atomic Model Theory Experiment It is also known as a hydrogen atom model or the Rutherford-Bohr Atomic Model Theory. 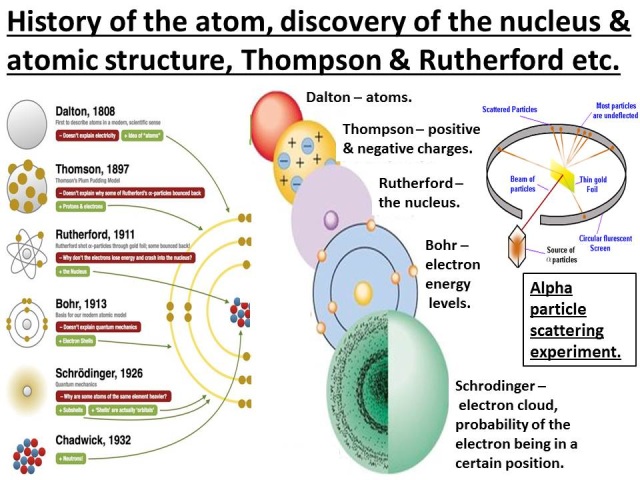
The Bohr atomic model theory states that atoms are composed of a nucleus, which consists of one or more protons and neutrons that are held together by nuclear forces. The electrons sit in energy levels around the nucleus, with the lowest possible energy level being electron number one and the highest being electron number eight. Atoms consist of a central nucleus, surrounded by electrons in orbital shells. In his model, Bohr postulated that electrons were placed in orbits that are referred to as orbitals. Niels Bohr’s atomic model was created based on previous research by Rutherford, Rutherford’s gold foil experiment, and Ernest Rutherford’s model of the atom. It was a postulation of Bohr that the electrons rotated in a circular orbit around the nucleus of the atom. The Niels Bohr Atomic Model theory is a model that was introduced by Niels Bohr in 1913 to describe the atom. What Was Niels Bohr Experiment? What Did Niels Bohr Discover? The atomic model is a theory that holds that the atoms in an element are different from one another and contain protons, electrons, and neutrons.īohr’s theory involved having electrons orbiting the nucleus in orbits and when they have near the nucleus and have an orbit with the same angular momentum, they could not change orbits without the release of some form of energy. Niels Bohr is a well-known Danish physicist that spent the majority of his life studying the atomic model. However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms.Niels Bohr Atomic Model Theory Experiment Niels Bohr Education & Life Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. 
When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. (Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. 
\): Bohr's atomic model hydrogen emission spectra.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
